Cymbalta Explained: Usage, Dosage, And Side Effects
Cymbalta (duloxetine) is an antidepressant and anxiolytic (anti-anxiety) medication used in the treatment of major depressive disorder (MDD), generalized anxiety disorder (GAD), fibromyalgia, and neuropathic pain (per WebMD and the FDA). It is also approved in Europe for the management of stress urinary incontinence (via Therapeutics and Clinical Risk Management).
Approved by the U.S. Food and Drug Administration (FDA) in 2004, Cymbalta became the heir to Prozac, just as the "game-changer" antidepressant's 20-year patent was set to expire (per Drugwatch). Cymbalta, and its generic form duloxetine, are used by an estimated 4.4 million patients, making it the 27th most prescribed drug in the United States (via ClinCalc).
In this article we'll take a deeper look into the usage, dosage, and side effects of Cymbalta.
The classes of antidepressants
As the name implies, antidepressants are a class of medications used in the treatment of depression (per MedlinePlus). According to the Centers for Disease Control and Prevention (CDC), 13.2% of American adults currently use antidepressant medications.
While antidepressants don't always cure depression, they can reduce symptoms (via the Mayo Clinic). According to InformedHealth.org, "The main aim of treatment with antidepressants is to relieve the symptoms of severe depression, such as feeling very down and exhausted, and prevent them from coming back. They are meant to make you feel emotionally stable again and help you to follow a normal daily routine."
There are a number of different classes of antidepressants (per the Mayo Clinic). These include the selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), atypical antidepressants, tricyclic antidepressants (TCAs), and monoamine oxidase inhibitors (MAOIs). As each of these classes of antidepressants act on specific neurotransmitters in your brain, each one will affect certain neurotransmitters in slightly different ways. Each class will also have unique side effects.
In deciding what antidepressant to prescribe, your healthcare provider will consider a number of factors. These may include your particular symptoms, possible side effects, whether a particular drug worked for a close relative, interactions with other medications, other health conditions, whether you are pregnant or breastfeeding, cost, and health insurance coverage.
Anxiolytics
With an estimated 31.1% of adults having experienced any anxiety disorder at some point in their lives, anxiety has become a very common issue affecting Americans (via the National Institute of Mental Health). Sometimes referred to as minor tranquilizers or anti-anxiety medications, anxiolytics are a class of drugs used to prevent or treat anxiety symptoms or disorders (per WebMD).
Benzodiazepines are the main class of medications used to relieve anxiety, according to Drugs.com. They act on a specific molecule called gamma aminobutyric acid (GABA) within the central nervous system to exert a calming effect. The main problem with the benzodiazepines is the high risk of dependence. Some of the most commonly prescribed benzodiazepines include alprazolam (Xanax), clonazepam (Klonopin), diazepam (Valium), and lorazepam (Ativan).
Another class of anxiolytics are the barbiturates. Due to the high risk of respiratory suppression, coma, and death, these drugs are not commonly used today. Other classes of drugs that also have an anxiolytic effect include the SSRIs, SNRIs (Cymbalta), TCAs, and buspirone (BuSpar). Many of these medications are commonly prescribed over benzodiazepines and barbiturates since they are unlikely to result in some of their unwanted effects. However, they may not work as quickly and may result in a temporary worsened state of anxiety after they are first started.
Differences between SSRIs and SNRIs
According to Medical News Today, selective serotonin reuptake inhibitors (SSRIs) and serotonin and norepinephrine reuptake inhibitors (SNRIs) are two commonly prescribed antidepressant medications.
SSRIs work by increasing the levels of serotonin in your brain, a neurotransmitter that helps the body regulate mood. Since the 1960s, several studies have shown that too much or not enough serotonin in the brain has been associated with causing depression (per Molecular Psychiatry). Although more recent research has found that this association may not be as valid as once thought, SSRIs are still the first line of treatment for depression and several other psychiatric disorders (via StatPearls). Some of the most commonly prescribed SSRIs include fluoxetine (Prozac), sertraline (Zoloft), paroxetine (Paxil), fluvoxamine (Luvox), citalopram (Celexa), escitalopram (Lexapro), and vilazodone (Viibryd).
According to PsychCentral, SNRIs increase (balance) levels of both serotonin and norepinephrine in the brain. Similar to serotonin, too much or too little norepinephrine has also been linked with mood dysregulation. Norepinephrine also plays several roles in the fight-or-flight response, sleep, attention, and memory (via Medical News Today). Because SNRIs increase levels of norepinephrine, they may also help patients who have difficulties with focus and concentration. Some of the most commonly prescribed SNRIs include venlafaxine (Effexor), desvenlafaxine (Pristiq), duloxetine (Cymbalta), milnacipran (Savella), and levomilnacipran (Fetzima) (per StatPearls).
Cymbalta can be used to treat major depressive disorder (MDD)
According to the Mayo Clinic, "Depression is a mood disorder that causes a persistent feeling of sadness and loss of interest." Sometimes referred to as major depressive disorder (MDD) or clinical depression, depression affects an estimated 5% of adults (via the World Health Organization).
During episodes of depression, an individual may experience symptoms of depressed mood, energy loss, fatigue, loss of pleasure, weight changes, changes in appetite, sleep changes, suicidal thoughts, loss of confidence, feelings of guilt, and difficulty concentrating (per the British Medical Journal). Diagnosis is often made after completing a comprehensive physical examination, laboratory testing, and psychiatric evaluation based on the criteria listed in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5).
Treatment is often managed through the use of prescription medications and psychotherapy. According to the American Family Physician, Cymbalta is approved by the U.S. Food and Drug Administration (FDA) for the treatment of MDD. Based on a number of clinical trials, it has been found to be safe, effective, and well tolerated by patients. A recent review article published in Frontiers in Psychiatry highlighted that 26 research studies found Cymbalta to be superior over placebo or other antidepressants such as sertraline, fluvoxamine, venlafaxine, paroxetine, escitalopram, fluoxetine, and bupropion.
Cymbalta can be used to treat generalized anxiety disorder (GAD)
Generalized anxiety disorder (GAD) is a mental health disorder characterized by excessive fear, worry, and a constant feeling of being overwhelmed (per StatPearls). Individuals with this condition typically experience excessive, persistent, and unrealistic worry about everyday things.
According to the DSM-5, GAD is diagnosed based on specific criteria. In order to be diagnosed with GAD, an individual must have excessive anxiety and worry for at least six months, they must have difficulty controlling the worrying, and they must be experiencing at least three of the following symptoms for at least six months: restlessness, fatigue, difficulty concentrating, muscle tension, sleep disturbance, and irritability.
SSRIs and SNRIs (such as Cymbalta) are the current first-line treatments for GAD (via Expert Opinion on Pharmacotherapy). Several clinical studies have shown that Cymbalta significantly improves symptoms of GAD (per the International Journal of General Medicine). Symptoms of GAD typically begin to improve within the first few weeks of treatment and any adverse effects tend to occur at relatively low rates.
According to CNS Neuroscience & Therapeutics, Cymbalta has also been suggested as a treatment option for other anxiety disorders such as panic disorder. Based on this study, Cymbalta treatment resulted in significant reduction in anxiety, as well as improvements in overall quality of life.
Cymbalta can be used to treat neuropathic pain
Neuropathic pain, sometimes referred to as nerve pain, occurs when there is damage or dysfunction of the nervous system (via the Cleveland Clinic). Because of this dysfunction, damaged nerve fibers may cause incorrect signals to be sent to pain centers in the brain, resulting in a variety of different types of pain. Spontaneous pain, or pain that occurs without any stimulation, may feel like a shooting, burning, or stabbing sensation with associated tingling and numbness. Evoked pain, also known as allodynia, is a form of pain that is caused by normally non-painful stimuli. There are a number of other types of neuropathic pain which include hyperalgesia, hypoalgesia, and dysesthesia.
Some of the most common causes of neuropathic pain include diabetes, alcoholism, multiple sclerosis, cancer, viral infection, and nerve damage following surgery or trauma (per the Brain & Spine Foundation). The primary strategy when it comes to treating neuropathic pain is to focus on reducing the pain. The most common treatment options include antiepileptics, antidepressants (including Cymbalta), opioids, injectable anesthetics or steroids, nerve stimulation therapy, and acupuncture.
In a 2009 study, scientists found "moderately strong evidence" that Cymbalta was effective in treating neuropathic pain (via Cochrane Reviews). Another study published in the journal Neurology found that Cymbalta taken either daily or twice daily was safe and effective in treating neuropathic pain caused by diabetes.
Cymbalta to treat fibromyalgia
According to the Mayo Clinic, fibromyalgia is a chronic disease that results in widespread musculoskeletal pain as well as a number of other symptoms including fatigue, sleep, memory loss, and mood issues. Fibromyalgia is more common in women and most often occurs following physical trauma, surgery, infection, or significant psychological stress. Fibromyalgia can also coexist with other conditions such as irritable bowel syndrome, chronic fatigue syndrome, anxiety, depression, and temporomandibular joint (TMJ) dysfunction.
Although there is no cure for fibromyalgia, there are a number of medications and non-drug therapies that can be used to help manage symptoms. People with fibromyalgia may benefit from cognitive behavioral therapy (CBT), physical exercise, acupuncture, as well as chiropractic and massage therapy (per the American College of Rheumatology).
The two drugs used to manage pain in fibromyalgia patients are Cymbalta and milnacipran (Savella). According to the journal Expert Review of Clinical Immunology, Cymbalta was shown to effectively diminish pain and increase physical function in several patients with fibromyalgia. Additionally, clinical trials have found that patients who saw pain relief did not experience any of the antidepressant or anxiolytic effects of Cymbalta (per Drugs of Today).
Cymbalta for stress urinary incontinence (SUI)
Stress urinary incontinence (SUI) is a condition affecting the urethra (tube that allows urine to leave the bladder) that results in urine leaking out, often during activities that increase abdominal pressure (via the Urology Care Foundation). SUI is a very common condition which affects an estimated one in three women over the age of 60.
In people with SUI, any forceful activity such as sneezing, laughing, coughing, or heavy lifting can put pressure on the bladder and cause a small amount of urine, often a few drops, to leak out. This condition often results from weakened, stretched, or damaged pelvic floor muscles. Some of the more common risk factors for SUI include pregnancy and childbirth, obesity, smoking, chronic coughing, and prostate surgery in males.
There are a number of treatment options for SUI (via StatPearls). In addition to making certain lifestyle changes, patients can undergo behavioral, pharmacological, or surgical management. Some behavioral methods include pelvic muscle exercises, bladder retraining, and electrostimulation. There are also a number of medications that can be used to help manage symptoms. These include oxybutynin, TCAs, topical estrogen, and Cymbalta (only approved in Europe), according to the Canadian Medical Association Journal).
According to Reviews in Urology, Cymbalta works to prevent urine leakage in SUI by increasing nerve stimulation to the muscle fibers surrounding the urethra (urethral sphincter muscle). In three clinical trials, patients who took Cymbalta had at least a 50% decrease in the frequency of their SUI episodes. In addition, these patients had significantly improved quality of life scores following treatment.
The dosage
According to the FDA, Cymbalta is available in 20 milligrams, 30 milligrams, and 60 milligrams delayed-release capsules. In contrast to immediate-release, delayed-release drugs are designed to release the active ingredient at a later time (per GoodRx Health). This allows more control over where the drug is released in the body. This also helps to prevent the medication from being broken down too early or to helps it reach the area requiring treatment.
The FDA mentions that "Cymbalta should be swallowed whole and should not be chewed or crushed, nor should the capsule be opened and its contents be sprinkled on food or mixed with liquids."
The recommended dosage someone should take depends on the specific condition being treated. Patients with major depressive disorder (MDD) are typically started on a dose of 40 to 60 milligrams per day, with the maximum dose being 120 milligrams per day. The starting dose for patients with generalized anxiety disorder (GAD) is 60 milligrams per day, with a maximum dose of 120 milligrams per day. Patients with diabetic peripheral neuropathic pain are placed on a dose of 60 milligrams per day. Patients with fibromyalgia are given a starting dose of 30 milligrams per day.
Dosages may be different for geriatric patients and those under the age of 17, as well as patients with hepatic or severe renal impairment (via Drugs.com).
Cymbalta may worsen depression
According to PsychCentral, it's possible that antidepressants can sometimes cause or worsen pre-existing depression. Specifically, "In October 2004, the Food and Drug Administration (FDA) placed a black box warning on antidepressant medications after research studies found an increased risk of suicidal thoughts and behaviors among children and teens. The FDA issued the same warning to young adults 2 years later."
These decisions were made after multiple clinical trials found patients with major depressive disorder (MDD) had increased suicidal ideations and behaviors when on these medications. However, the exact biological mechanism behind how this happens remains unclear. Also, it may be difficult to distinguish whether it is the antidepressants causing the suicidal thoughts, if a patient's depression is not being adequately treated, or if a patient is simply not responding to the medication.
On the FDA's prescribing label for Cymbalta, the black box warning (on page three) states that, "Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of Cymbalta or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need."
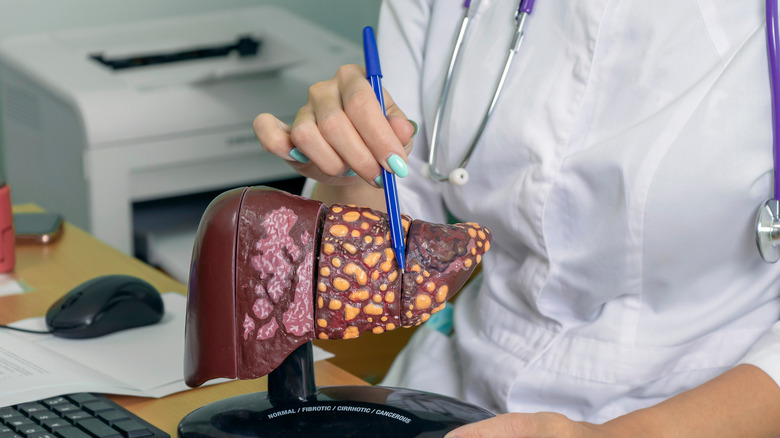
Cymbalta may cause liver inflammation
Another rare but potentially serious adverse effect of Cymbalta is hepatotoxicity (per the FDA). According to a report published by the National Institute of Diabetes and Digestive and Kidney Diseases, an estimated 1% of individuals taking Cymbalta had liver test abnormalities. Specifically, lab results in these patients showed that levels of a specific liver enzyme called ALT were three times the upper normal limit. Most of these elevations resolved over time and did not require any changes to the dosage or discontinuation of Cymbalta.
However, there have been rare instances of liver injury, acute liver failure, and chronic hepatitis which occurred within one to six months after starting Cymbalta. The exact mechanism behind the liver inflammation is not known, but likely has to do with one of the metabolic byproducts of the drug after it is metabolized by the liver. Individuals who do not tolerate Cymbalta may also have similar reactions to other SNRIs and SSRIs. These individuals should be carefully monitored when switching to one of these other medications.
According to the FDA's prescribing label for Cymbalta, "Hepatic failure, sometimes fatal, has been reported in patients treated with Cymbalta. Cymbalta should be discontinued in patients who develop jaundice or other evidence of clinically significant liver dysfunction and should not be resumed unless another cause can be established."
Other side effects of Cymbalta
Other common and less serious side effects seen in patients taking Cymbalta include asthenia (weakness), constipation, drowsiness diarrhea, dizziness, fatigue, hypersomnia (trouble staying awake during the day even if you sleep well at night), insomnia, headache, nausea, sedation, and xerostomia (dry mouth), according to Drugs.com. Of these, the most commonly reported include nausea, drowsiness, headache, and dizziness.
A number of post-marketing reports also identified a number of rare but possible side effects that may be caused by Cymbalta. These included reports of pancreatitis, abnormal bleeding, renal dysfunction, and arrhythmias.
Another scenario in which patients taking Cymbalta can experience adverse effects is if they abruptly stop taking the drug (via the Journal of Affective Disorders). Abrupt discontinuation of Cymbalta can result in dizziness, nausea, headache, paresthesia (abnormal tingling or pricking sensation), vomiting, irritability, and nightmares. Similar adverse effects are also seen in patients who suddenly stop taking other SNRI or SSRI medications. For this reason, it is common practice for healthcare providers to gradually reduce the dose of Cymbalta (as well as other SSRI or SNRI drugs) no less than two weeks before stopping the treatment.
Those who shouldn't take Cymbalta
Cymbalta is not recommended for patients with certain underlying medical conditions (via the FDA). One such contraindication for Cymbalta is patients who are taking or have recently stopped taking monoamine oxidase inhibitors (MAOIs). The MAOIs are an "extremely strong class of antidepressants" with a large side effect profile and a number of additional safety concerns (per WebMD). If this contraindication is not strictly followed, patients can be at risk of developing serotonin syndrome, a potentially life-threatening condition that requires immediate medical attention (via Psychiatry Online and WebMD).
According to the FDA, another major contraindication for Cymbalta is in patients with uncontrolled narrow-angle glaucoma. Glaucoma is a disease in which increased pressure inside the eye causes damage to the optic nerve, resulting in vision loss (per the Optometrists Network). Some cases of glaucoma, such as drug-induced acute angle closure glaucoma (AACG) may result in blindness (via the Journal of Current Glaucoma Practice). Because of this risk, as well as numerous reports of AACG having been associated with the use of SNRIs, including Cymbalta, uncontrolled narrow-angle glaucoma is one of the major contraindications for this medication.
Also mentioned in the FDA's prescribing label for Cymbalta is that pregnant and nursing mothers should only use this medication if its potential benefits justify the potential risks to the fetus or child.